A Brief Colonial History Of Ceylon(SriLanka)
Sri Lanka: One Island Two Nations
A Brief Colonial History Of Ceylon(SriLanka)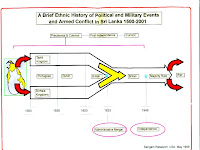
Sri Lanka: One Island Two Nations
(Full Story)
Search This Blog
Back to 500BC.
==========================
Thiranjala Weerasinghe sj.- One Island Two Nations
?????????????????????????????????????????????????Friday, May 26, 2017
Modi's generics-only drugs plan worries health experts, Indian pharma sector
A
patient holds free medicine provided by the government at Rajiv Gandhi
Government General Hospital (RGGGH) in Chennai July 12, 2012. REUTERS/Babu/Files
India's plan to bring in a law to ensure doctors prescribe medicines
only by their generic names risks proliferating the sale of substandard
drugs in a country where regulation is already lax, doctors and
pharmaceutical executives say.
Prime Minister Narendra Modi said last month that the government was
looking at such a law to improve affordability of medicines in the
country of 1.2 billion people, where the majority live on less than $2 a
day.
The government has not given any details or timeframe for the planned
legislation but some in the industry fear that hasty execution could
harm, not help, public health.
Large drugmakers in India such as Cipla and Sun Pharmaceutical
Industries that mostly sell generic drugs under their own brand name, or
'branded generics', compete with much smaller garage manufacturers,
many of which operate with scant quality checks.
It is a fiercely competitive, high-volume market, dominated by the big
players like Cipla, who employ armies of salesmen to sell their drugs.
Some doctors say they tend to prescribe branded generics to patients
because they are confident of their quality.
While the government wants to make cheaper non-branded drugs available
to consumers, pharmaceutical executives said any law would have to
stipulate that the drugs consumers get from pharmacists meet certain
quality standards.
"I think the quality aspect is very important for the government to
address ... to make sure that all companies in India are on the same
quality footing," said Cipla's CEO Umang Vohra.
There is no national data comparing the quality of branded generics,
versus unbranded ones. But small studies have shown that unbranded
drugs, typically procured by the government for the public health
system, have quality issues.
A report by the country's federal auditor in 2012 showed that 31 percent
of drugs procured by the government for the Armed Forces Medical Stores
were substandard in 2010-11, up from about 15 percent in 2006-07.
Results of the government's largest-ever national survey to test drug
quality showed this year that roughly 10 percent of the drugs in the
government supply chain were not of standard quality, versus 3 percent
of drugs available at pharmacies, which are usually branded generics.
"Generics are fine, but there has to be a proper rigorous mechanism to
enforce quality, like the U.S., and unless India evolves on that it will
be disastrous," said Nilesh Gupta, managing director of Lupin, India's
No. 3 drugmaker by sales.
IMPRACTICAL
Substandard medicines range from drugs that don't work at all to those
that don't work as expected, thereby contributing to antimicrobial
resistance - a major global health problem.
"The idea is very good, but the government needs to ensure the
availability of good quality generics," said Vijay Panikar, a
Mumbai-based diabetologist.
Indian drugmakers' manufacturing standards, including those of big
companies such as Sun Pharma, have been found to have fallen short of
international standards in recent years. An industry official said last
week that it will be at least five years before Indian manufacturing and
data reliability meet the standards of the United States, India's
biggest drugs export market.
"It's a work in progress ... I think the bigger companies understand the
problem and are working to fix it," said Gupta of Lupin.
Health experts warn of a series of problems with the implementation of a
generics-only rule. For example, half the Indian market is made up of
combination drugs, and it would be impractical to ask doctors to
prescribe a series of chemical names, said S. Srinivasan, a doctor and
member of the People's Health Movement, a New Delhi-based NGO.
Doctors opposed to the planned new rule warn it would put too much power in the hands of the chemists.
"Today if I write a generic, the chemist will decide which drug to give,
and he will obviously give the one in which he has the biggest margin,
without caring about quality," Panikar said.
(Reporting by Zeba Siddiqui in Mumbai; Editing by Euan Rocha and Susan Fenton)


